Executive Summary
There is a notion that the drug pricing proposals in the House-passed Build Back Better Act (BBBA) are extra modest and fewer threatening to medical innovation than these of the draconian H.R. 3 proposal. Policymakers, the media, and the public ought to perceive that it is a deeply flawed impression. This perception summarizes the BBBA’s key drug coverage provisions and examines their potential impacts. It finds that the BBBA’s drug insurance policies would dramatically curtail future innovation and imperil the financial advantages the United States derives from the biopharmaceutical sector.
Key Points:
- The BBBA would set up an specific authorities price-setting regime for prescription drugs, reaching into all corners of the U.S. well being sector, each private and non-private.
- The BBBA doesn’t set up a real “negotiation” of drug costs in Medicare; reasonably it could empower the secretary of Health and Human Services (HHS) to dictate costs to producers who would have little to no leverage.
- The BBBA would enshrine a singular and punitive 95 % excise tax on gross income of a remedy if the producer doesn’t conform to the secretary’s calls for.
- The BBBA would set up a ceiling worth, however not like previous proposals there isn’t any ground worth under which the secretary could be unable to drive additional concessions.
- Under the BBBA, for the first time, the federal authorities would cap the worth of all medicine all through the complete well being care system by prohibiting any drug’s worth from growing quicker than the charge of inflation.
- The BBBA proposals would hurt shoppers by decreased innovation and better launch costs for medicine and therapies.
- One latest research discovered that the BBBA would end in 135 fewer new drug approvals, 188 fewer new indications for present therapies, and a discount of 331.5 million life years amongst sufferers.
- The mixture of worth setting by the HHS secretary and inflation penalties would doubtless cut back generic and biosimilar market entry, placing in danger potential financial savings and improved remedy choices for tens of millions of Americans.
- Price controls will basically remove future enhancements in insulins.
- BBBA’s inflation penalties are prone to end in larger launch costs and will drive worth will increase commensurate with inflation for therapies whose costs wouldn’t improve beneath present regulation.
- The BBBA would hurt and endanger the expansive financial exercise generated by the biopharmaceutical trade in the United States.
- The BBBA would scale back biopharmaceutical trade income by $2.9 trillion by 2039 and places in danger a U.S. biopharmaceutical sector that generates greater than $1 trillion in financial exercise yearly, employs greater than 800,000 staff, and helps greater than 4 million jobs throughout the U.S. economic system.
- In the mid-Eighties, as European nations imposed stringent worth controls, Europe noticed a flight of funding in drug growth and manufacturing to the United States. Under the BBBA, the United States would threat an analogous loss in competitiveness to nations comparable to India and China which can be aggressively in search of to bolster their very own biopharmaceutical sectors.
- There are higher methods to decrease drug costs than the BBBA.
- Congress ought to discard the the rest of the BBBA and deal with pursuing bipartisan reforms to Medicare Part D, just like the redesign included in the BBBA.
- Legislative modifications to drug rebates, like these pursued by rulemaking beneath the Trump Administration, would make sure that sufferers with excessive drug prices obtain significant help.
- The single greatest option to convey down costs is to extend provide and competitors; policymakers ought to take away regulatory obstacles to new therapies and follow-on merchandise coming to market, whereas additionally working to make sure enforcement of legal guidelines concentrating on anticompetitive practices.
Introduction
Annual well being care spending in the United States elevated 9.7 % in 2020, totaling $4.1 trillion. While the COVID-19 pandemic has performed a job in latest will increase, well being care spending and prices have each been rising quickly for years. In 2020, 19.7 % of the U.S. economic system was dedicated to well being care spending.[1] Given these developments, policymakers throughout the political spectrum have lengthy been involved with constraining well being care value development. But whereas conservatives have sought to deal with this coverage problem by selling selection and competitors in the non-public market, progressives have centered on decreasing income for well being care firms and growing the federal authorities’s function in offering and paying for care. Most notably, progressive politicians and activists have lengthy been keen to focus on the income of pharmaceutical producers.
Prescription medicine, nonetheless, make up a comparatively small share of complete well being care spending. According to the Centers for Medicare and Medicaid Services (CMS), retail prescription drug spending made up simply 8.4 % of all U.S. well being care spending in 2020.[2] While these figures don’t account for therapies administered by suppliers in hospitals, nursing houses, or related settings, the consulting agency Altarum estimates that non-retail prescription drug spending accounts for 4.5 % of total well being care expenditures yearly.[3] Though complete spending on prescription therapies will not be a big share of total well being spending, sufferers usually tend to bear the value of remedy straight on account of the excessive coinsurance and deductibles more and more frequent to drug protection. Concern over the growing value of many biopharmaceutical therapies is subsequently bipartisan, however progressives have constantly pursued insurance policies that will constrain future innovation and limit American sufferers’ entry to the broadest vary of therapies.
Much ink has been spilled over the apparent flaws of the Elijah E. Cummings Lower Drug Costs Now Act (H.R. 3), Speaker Pelosi’s laws aimed toward constraining prescription drug costs, however far much less consideration has been given to the drug insurance policies that have been included in the House-passed Build Back Better Act (BBBA).[4] In reality, there’s a widespread notion that, as a result of H.R. 3 was not included verbatim in the BBBA, the BBBA’s drug worth provisions are much less egregious and pose far much less threat to shoppers and the U.S. biopharmaceutical sector. This understanding is inaccurate.
This paper summarizes the BBBA’s key drug coverage provisions and examines their potential impacts. It finds that the BBBA’s drug insurance policies would dramatically curtail future innovation and would imperil the financial advantages the United States derives from the biopharmaceutical sector.
Key Drug Pricing Policies in the BBBA[5]
While motion on the BBBA has stalled in the Senate, the House-passed model of the laws accommodates a quantity of drug-related insurance policies, specifically, Medicare negotiation of drug costs, inflation penalties, new regulation of insulin merchandise and costs, and reforms to the Medicare Part D prescription drug profit.
Medicare Negotiation for Drug Prices
Under the BBBA, starting in 2025, the secretary of Health and Human Services (HHS) could be licensed to “negotiate” the costs of as much as 10 “negotiation-eligible drugs.” In 2026 and 2027, the cap will increase to fifteen medicine yearly, and rises to twenty medicine in 2028 and past. Part B medicine—these medicine administered by a medical supplier in a hospital, nursing dwelling, or related setting—could be exempt till 2027. Additionally, all insulin merchandise would robotically be obtainable for negotiation past the yearly caps.
A negotiation-eligible drug is outlined as a small-molecule or biologic (together with licensed generics) remedy that has had Food and Drug Administration (FDA) approval for a minimum of seven years for a small-molecule drug or 11 years for a biologic that’s amongst the 50 single-source medicine with the highest complete expenditures in Part B or Part D. Orphan medicine or “low-spend” medicine are excluded, with low-spend being outlined as a drug or biologic on which Medicare spends lower than $200 million yearly (adjusted by the shopper worth index in future years). The decreased costs could be efficient after a further two years, which means small-molecule medicine would have costs decreased 9 years after approval and 13 years for biologics.
The BBBA would set a ceiling for negotiated worth of between 40-75 % of the non-federal common producer worth (AMP)—the common worth paid by wholesalers, internet of immediate pay reductions—cutting down relying on how far the drug is previous its preliminary exclusivity interval. There would, nonetheless, be no ground under which HHS couldn’t demand worth concessions. Unlike H.R. 3, the negotiated worth wouldn’t be straight utilized to the non-public well being care market, however the negotiated charge or “maximum fair price” could be publicized.
In order to offer the secretary with leverage in negotiations, the laws would set up an excise tax particularly on gross sales of medicine the secretary has focused for negotiation however for which the producer has not agreed to the secretary’s goal worth. The excise tax could be utilized for any interval through which the producer is in “non-compliance.” The tax would begin out at 65 % of gross sales of the remedy for the first 90 days of non-compliance, growing at common intervals till topping out at 95 % for any interval of non-compliance past 270 days.
Inflation Penalties
The BBBA would set up penalties for drugmakers in the event that they improve the worth of a selected remedy quicker than the charge of inflation. For medicine coated by Part D, the AMP could be assumed as the base worth of the drug for the objective of monitoring worth will increase. A drug’s AMP could be benchmarked to October 2020, whereas inflation could be benchmarked to September 2021. Drugmakers may nonetheless improve their costs above inflation, however they must write a verify for the distinction. For instance, if a drug’s AMP was $110 per unit in October 2020, and the inflation-adjusted AMP in 2023 is $120, however the precise AMP is $130, the producer must pay the authorities $10 for each unit offered in 2023. The Part D inflation cap would apply to all medicine with a worth of greater than $100 per affected person, per yr.
In Part B, the precept is essentially the identical with a couple of variations. The worth of the drug to be thought-about could be the Average Sales Price (ASP). ASP could be benchmarked to July 2021, whereas inflation could be benchmarked to September 2021. While each penalties would take impact in 2023, the Part B penalty could be assessed quarterly, whereas the Part D penalty could be paid yearly. The penalty could be utilized to all single-source medicine in Part B with prices exceeding $100 per affected person, per yr—and biologics would nonetheless be thought-about single supply even when there have been biosimilar rivals. Biosimilars would even be topic to penalties if their worth is above that of the reference product.
Insulin Price Restrictions
The House-passed BBBA particularly targets insulin costs, making all insulin merchandise robotically topic to Medicare negotiation. The BBBA would additionally unilaterally restrict cost-sharing for insulin by Part D to $35 monthly.
The BBBA would intercede in the group and particular person insurance coverage markets to restrict affected person insulin prices. Starting in 2023, well being insurers providing group or particular person medical insurance protection could be required to offer protection for a minimum of one of every insulin dosage kind (vial, pump, or inhaler) of every kind of insulin (rapid-acting, short-acting, intermediate-acting, long-acting, and premixed). Further plans could be required to restrict affected person prices for insulin to not more than both $35 for a 30-day provide, or an quantity equal to 25 % of the negotiated worth of the insulin product for a 30-day provide—internet all worth concessions—whichever is decrease.
Medicare Part D Reforms
The House-passed BBBA undertakes a big redesign of the Medicare Part D program, starting in 2024, aimed toward realigning plan and producer incentives to constrain drug costs and to restrict beneficiaries’ out-of-pocket (OOP) prices. The broad framework of the proposal—initially outlined by the American Action Forum’s (AAF) Tara O’Neill Hayes in 2018—has garnered bipartisan assist, though there have been partisan variations over some of the particulars.
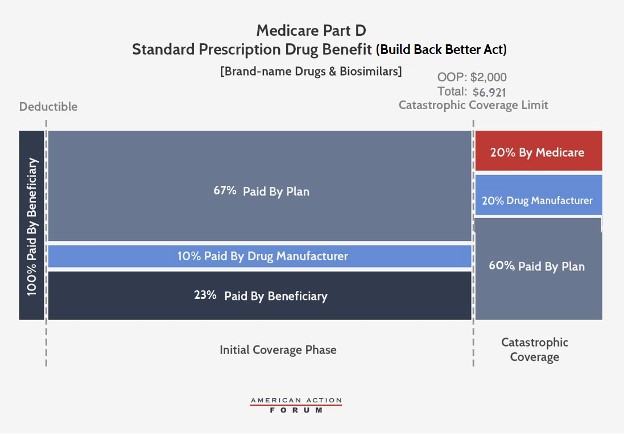
Under the BBBA, brand-name and biosimilar drug producers could be accountable for 10 % of prices in the preliminary protection part and 20 % in the catastrophic part. Government reinsurance would fall to twenty % for brand-name medicine and biosimilars and to 40 % for generic medicine. Insurer legal responsibility in the catastrophic part would improve to 60 % for all medicine. The catastrophic part would start at $2,000 in OOP prices, capping beneficiary prices at that time.
The BBBA would additionally cut back beneficiaries’ coinsurance legal responsibility to 23 % in the preliminary protection part (from 25 % at present) and their premium legal responsibility to 23.5 % (from 25.5 % at present). Consequently, the federal premium subsidy charge would rise to 76.5 % (from 74.5 %) and insurer legal responsibility in the preliminary protection part could be 77 % for generic medicine and 67 % for brand-names and biosimilars.
Last, the BBBA would enable for beneficiaries’ OOP prices to be “smoothed” over the course of the yr, reasonably than doubtlessly having to pay as a lot as $2,000 in a single month.
While the BBBA model of the Part D redesign retains the 10 % producer share in the preliminary protection part that was added in the H.R. 3 model of the proposal, the laws would decrease the producer share in the catastrophic part from 30 % in H.R. 3 to twenty %, whereas AAF initially proposed 9 % (notice that AAF used 9 % as a result of that was decided to be the charge at which pharmaceutical firms could be liable for the identical degree of prices at the time the authentic evaluation was achieved, whereas AAF was impartial on whether or not producers’ share of prices ought to improve).
Negative Impacts of the BBBA’s Drug Policies
The declare that that drug costs are larger than they fairly ought to be is extensively accepted. Progressive policymakers and activists specifically are fast to argue that drugmaker income are larger than acceptable, or that producers ought to solely be capable to pocket a “reasonable”—however undefined—revenue. Part of the subject is that many don’t acknowledge the excessive threat related to pharmaceutical growth. On common it takes greater than a decade to convey a drug to market, and most therapies by no means get there. Between 2011 and 2020, solely 7.9 % of compounds that began Phase 1 scientific trials made it to market.[6] Calculating how a lot was spent on a selected drug’s growth after which calculating a “fair” share markup for revenue fails to account for funding in unsuccessful analysis efforts, or the inherent threat buyers take once they present analysis and growth (R&D) capital to innovators. An inexpensive return on funding (ROI) will look totally different if the threat of failure is larger, and buyers anticipate a better ROI in trade for the dangers associated to pharmaceutical growth relative to different funding choices. In the absence of enough ROI, enterprise capital for pharmaceutical innovation will develop into more and more scarce.
While the Part D redesign proposal in the BBBA does have a measure of bipartisan assist, and goals to appropriate perverse incentives inside the program, the total affect of the mixed insurance policies could be to strangle innovation, restrict entry to new therapies for U.S. sufferers in the future, and endanger the financial advantages of a vibrant U.S.-based biopharmaceutical sector.
Overall Impact on Innovation
At the most elementary degree, any insurance policies that cut back pharmaceutical trade income can have downward stress on future innovation. In a latest paper from the University of Chicago, Tomas Philipson and Troy Durie estimate {that a} 1 % discount in pharmaceutical trade income leads on common to a 1.54 % drop in R&D spending.[7] This doesn’t imply that any coverage that reduces trade income is inherently misguided, however policymakers have to be cognizant about the potential impacts of the insurance policies they advance. Further, punitive insurance policies aimed primarily at decreasing drug firm income—reasonably than these addressing particular market failures or perverse incentives enshrined in present regulation—can have unfavourable long-term impacts far outweighing any short-term satisfaction derived from “sticking it” to producers.
AAF researchers have documented the potential affect on innovation of earlier insurance policies aimed toward decreasing pharmaceutical costs, particularly the Trump Administration’s International Price Index (IPI) proposal, and Speaker Pelosi’s H.R. 3. These proposals differ notably from the BBBA in that they tied worth controls to the worth of a drug in designated international nations. As such, these analyses can’t be straight utilized to the BBBA, however they do present some context for the potential affect of this laws’s worth controls.
According to evaluation by AAF’s O’Neill Hayes in 2019, feedback to CMS on the aborted IPI proposal, if that demo had been utilized to all Part B medicine—expenditures for which equal roughly $30 billion—trade revenues would have been decreased roughly $9 billion per yr. Considering that the value of efficiently bringing a drug to market has been estimated at roughly $2.87 billion, the $9 billion in misplaced income per yr doubtlessly attributable to the IPI proposal could be equal to the value of three new medicines annually, or 30 fewer new therapies over 10 years. In the case of H.R. 3’s Average International Market (AIM) worth, drug costs could be capped at 120 % of the index, reasonably than 126 % in the IPI proposal, and the capped worth could be utilized to all U.S. payers reasonably than restricted to Medicare Part B, which accounts for under 10 % of all drug expenditures in the United States. [8] If the impact on drug growth of the AIM worth have been just like the affect of the IPI, increasing these results to 100% of the U.S. market could be the equal of 30 fewer medicine per yr or 60 % of the complete quantity of new medicine permitted by the FDA in 2021.[9] Extrapolated over 10 years, H.R. 3 would have doubtlessly decreased trade income by the equal value of 300 new therapies. Of course, these proposals could be unlikely to end in dollar-for-dollar reductions in R&D, so the precise quantity of misplaced therapies could be decrease. These estimates are additionally in a roundabout way relevant to the BBBA as a result of, whereas these previous insurance policies limit drug costs to a restricted vary primarily based on established worldwide costs, the BBBA would implement a system of open-ended and steep worth concessions primarily based on home costs and enforced by a staggering 95 % tax on gross income of a selected remedy when a producer fails to satisfy HHS’ worth calls for.
Philipson and Durie, in a sturdy evaluation of the BBBA provisions revealed November 2021, estimate the laws would scale back trade income by an astronomical $2.9 trillion by 2039. They attribute $1.77 trillion to the inflation rebates, $986.9 billion to authorities “negotiation,” and $138.1 billion to the Part D reforms. Using their estimates of the affect of income reductions on R&D spending, the authors calculate that the BBBA would end in 135 fewer new drug approvals by 2039, and that additional disincentive to researching extra indications will result in 188 fewer new indications for present therapies over the identical interval. The authors additionally estimate that the insurance policies would end in 331.5 million fewer life years by 2039. Significantly, the authors solely apply the inflation limits to Medicare, however the inflation penalties will restrict pricing in the non-public market as effectively, resulting in even larger impacts on future innovation. They additionally assume that costs will probably be set at the absolute highest quantity allowed beneath the BBBA, however there isn’t any worth ground, and the HHS secretary would have substantial leverage to drive worth concessions effectively under the most worth.
Rather than being extra restricted in its affect on innovation than earlier drug pricing proposals, the BBBA’s deleterious results could be a minimum of similar to previous proposals comparable to H.R. 3, and doubtlessly even bigger.
It also needs to be famous that the BBBA’s heavy intervention in the insulin market, standard although these provisions are prone to be, will nearly actually finish substantive innovation round insulin merchandise and supply mechanisms, as there will probably be little monetary incentive for firms to proceed to put money into their growth.
Medicare Negotiation: Dressed up Price-Fixing
Democrats of their political messaging have lengthy touted the must “let Medicare negotiate drug prices.” The concept that the federal authorities would make no effort to get the greatest deal attainable on a service or good intuitively feels unsuitable—although it’s sadly comparatively frequent throughout federal packages—a lot in order that even former President Trump embraced the left’s rhetoric and known as for Medicare to barter drug costs. This messaging, nonetheless, misstates the actuality of how Medicare pays for medicine. One would possibly suppose from these speaking factors that no negotiations happen between the Medicare prescription drug program and drug producers. In reality, the Medicare Part D program has strong negotiation and competitors constructed into its very cloth.[10] Insurance firms providing drug protection by Part D negotiate straight with producers to get the greatest worth they will for the medicine they supply. Getting a lower cost advantages the prescription drug plan straight and permits it to decrease premiums to draw seniors. In this fashion, the negotiations drive down premiums, copays and total drug prices. Plans are capable of drive reductions by providing most well-liked placement on their formularies to particular therapies in trade for decrease costs. In some instances—with the exception of particular protected courses of medicine—a plan would possibly decline to cowl a selected remedy in any respect as half of its negotiations. This could be an issue if there have been just one formulary for all beneficiaries, however beneficiaries are in a position to decide on between a variety of plan choices, permitting them to pick out a plan that most closely fits their wants. In 2022, the common Medicare beneficiary can have a selection of 23 stand-alone Part D plans, and 31 Medicare Advantage plans that embrace drug protection.[11]
To defend this aggressive atmosphere, federal regulation prohibits the HHS secretary from interfering in the negotiations between plans and producers. The Congressional Budget Office (CBO) has lengthy held that merely eradicating this “noninterference” clause wouldn’t end in any financial savings for the program as a result of the secretary has no beneficiaries to barter on behalf of, and no leverage for driving worth concessions.[12] Giving the secretary the authorized authority to barter straight with producers will both end in a single negotiated worth for every drug—which can then need to be accepted by all insurers—thus undermining the aggressive construction of Part D, or it can end in nothing.
The authors of the BBBA have opted for the former, permitting the secretary to set costs that will probably be utilized to all plans, and giving the secretary leverage to drive worth concessions by way of the draconian 95 % excise tax on gross income for any remedy whose producer is unwilling to satisfy the secretary’s worth calls for. Setting apart the unfavourable long-term results of the strict worth controls envisioned by the BBBA, proponents ought to dispense with the fiction that this could resemble something remotely like a negotiation. Given the 95 % excise tax—the like of which seems nowhere else in federal regulation—the secretary could be free to wield in opposition to noncompliant innovators, “price extortion” could be a extra sincere label for this provision than “price negotiation.”
Ramifications of Inflation Penalties
While the BBBA wouldn’t apply Medicare’s negotiated costs for medicine to non-federal packages, the most important implication of the BBBA’s dollar-for-dollar penalties on worth will increase that exceed the charge of inflation is that, for the first time, the federal authorities could be unilaterally capping drug costs nationwide, each in federal packages and in the non-public market. This shift in the federal authorities’s posture towards non-public markets, negotiations, and competitors can’t be overstated.
Additionally, the BBBA units the inflation benchmark to a later date than the worth benchmark. As a outcome, the laws extracts extra income from drugmakers to pay for the BBBA’s different provisions—reinforcing the declare that a minimum of half of the functions of the drug provisions is solely to generate cash to pay for unrelated new spending. If the coverage benchmarked each drug worth and inflation to September 2021, CBO would doubtless have assumed that drugmakers would preserve their worth will increase to the charge of inflation. There could be financial savings on account of slower worth development over time, however they wouldn’t be large. By capturing the latest inflation spike and back-dating drug costs far sufficient to make sure that pricing selections already made are topic to the new coverage, Democrats ensured that the CBO rating included extra income from penalties, and that producers must pay for Democrats’ final yr of inflationary spending insurance policies.
Faced with restrictions on future pricing flexibility, drugmakers could be incentivized to extend preliminary launch costs in response to inflation penalties. While these merchandise would ultimately be topic to HHS’s price-setting regime, these compelled worth concessions wouldn’t take impact till years after the product’s launch, additional incentivizing producers to maximise preliminary income by larger launch costs.
It is price noting that the BBBA drug provisions could be launched in an atmosphere of normal worth inflation not seen in 4 many years. The imposition of worth controls on insulin and different medicine would assure that they are going to be underpriced in actual phrases in very quick order—a recipe for additional inefficiency and injury to innovation incentives. At the identical time, there could be medicine that can see their costs rise at inflation—as a result of the BBBA basically blesses such a worth rise as “legitimate”—though no such improve is merited on the fundamentals. The outcome could be costs which can be too excessive in actual phrases and a hurt to shoppers.
Economic Damage to the Biopharmaceutical Sector
The biopharmaceutical sector in the United States creates greater than $1 trillion in financial exercise and employs greater than 800,000 staff—at a median compensation over twice the nationwide common. More broadly, the trade helps greater than 4 million jobs throughout the U.S. economic system, and generated over $67 billion in federal, state, and native tax income in 2017 alone.
The BBBA’s insurance policies aimed toward decreasing trade revenues put this vibrant financial engine in danger. In 1986, R&D investments by pharmaceutical corporations in Europe exceeded R&D in the United States by roughly 24 %.[13] Following the imposition of authorities worth controls in lots of European nations, and consequently the decreased return on funding, R&D spending by pharmaceutical firms grew at an annual charge of simply 5.4 % in the European Union, in contrast with 8.8 % development in the United States. As such, greater than half of the world’s pharmaceutical R&D investments have been made in the United States since the flip of the century, whereas lower than 30 % has been invested in Europe.[14] While shifting patterns of funding are the product of many elements, traditionally R&D and manufacturing investments have moved away from nations through which strict worth management regimes are applied. With nations comparable to India and China, amongst others, aggressively in search of to bolster their very own biopharmaceutical industries, the BBBA would put in danger the financial advantages the United States derives from the sector and would benefit different nations of their efforts to lure away investments at present being made on this nation.
Impact on Generic and Biosimilar Market Entry
Another under-appreciated level of concern with the BBBA is the method the laws’s provisions may disincentivize future growth of generic and biosimilar therapies. Historically, flow-on merchandise have led to vital value financial savings for American sufferers and have been a main driver of prescription medicines’ comparatively small share of complete well being care expenditures. Ironically, the extra profitable the HHS secretary is in leveraging the BBBA’s punitive excise tax to drive worth concessions, the fewer generic and biosimilar merchandise are prone to come to market. Follow-on merchandise are capable of dramatically undercut name-brand medicine and biologics on worth as a result of they don’t have the identical R&D expenditures and since their decrease costs enable them to realize bigger market shares. But if the worth distinction between a name-brand drug, topic to the secretary’s worth controls, and a brand new generic is marginal and even non-existent, the potential of a generic to realize market share will probably be decreased.
It could also be that HHS is ready to drive enough worth concessions—at the value of future innovation—to offset some of the misplaced financial savings on account of a decimated generic and biosimilar pipeline, however decrease costs usually are not the solely profit of follow-on merchandise. Different sufferers reply in a different way to the identical remedy, so a sturdy pipeline of follow-on therapies ensures sufferers usually tend to have entry to a remedy with out undesirable unintended effects. Reduced market entry of generics and biosimilars may result in fewer choices for docs to assist sufferers keep away from antagonistic reactions and unintended effects.
Pharmaceutical Policy Options to Consider
While progressives spin their collective wheels making an attempt to focus on drugmaker income, there are some viable options for tackling drug costs in ways in which promote competitors and higher align the incentives inherent in federal regulation.
Drug Rebates
In 2019 the Trump Administration proposed vital modifications to the construction of drug rebates. While Congress has delayed and sought to repeal this rulemaking, it could be smart to rethink. Under present regulation, drug producers sometimes present vital rebates for medicine supplied at the pharmacy counter (averaging almost 30 % in Medicare Part D), particularly for medicine with competing options. These rebates are mostly paid to pharmacy profit managers (PBMs) in trade for most well-liked placement on the insurance coverage plan’s drug formulary. The PBMs, nonetheless, don’t often share these rebates straight with sufferers, as a substitute sometimes utilizing the rebates to carry down premium prices for everybody. But utilizing rebates on high-cost medicine to broadly decrease premiums as a substitute of passing them by to beneficiaries ends in the (high-cost) sick subsidizing the (low-cost) wholesome, which appears counter to the intent of an insurance coverage product.
The rebate rule, if applied, would change that apply. Drug rebates would not be allowed until they’re fully handed by to the affected person at the level of sale. This change would nearly actually result in elevated premiums Part D premiums, which is why there was opposition. Those will increase are prone to be minimal, nonetheless, as the value improve could be unfold throughout all beneficiaries. On the different hand, the decreased cost-sharing bills that the highest-cost beneficiaries would see ought to outweigh these premium value will increase, leading to a internet profit to sufferers. Those sufferers with the highest prices would see the biggest profit. The Trump Administration may solely suggest modifications to have an effect on rebates in the Medicare program by rulemaking; if Congress have been to enact a legislative model of the rebate rule, nonetheless, it may prolong the coverage all through the insurance coverage system, which is an method price consideration.
Competition and Increased Supply
History has confirmed the greatest option to cut back the worth of a superb for which there’s rising demand is to extend its provide by competitors. For drug pricing, which means bringing generics and biosimilars to market to compete with brand-name medicine.
A now-classic instance of this phenomenon is the Hepatitis C remedy, Sovaldi, which contributed over $3 billion to 2014 expenditures alone.[15] While the drug was fairly costly, you will need to notice two issues. First, Sovaldi—and its eventual rivals—supplied a remedy for what had been up till that time a costly-to-manage power illness. Second, as rivals joined the market, the worth of Sovaldi was lower in half. Where there’s competitors, costs come down. The FDA has been doing its half by approving a document quantity of generic medicine and biosimilars.[16] But different obstacles to unlocking strong market competitors stay.
Legal Enforcement of Competition Policy
Often, as soon as a generic drug has been on the market lengthy sufficient, it acquires sufficient of the market share that the brand-name producer stops producing its model of the drug. In many instances, the worth reaches a low sufficient level at which different generic rivals additionally exit the market, leaving a sole producer. In some high-profile instances we see what quantities to abuse of monopoly energy—that sole producer taking benefit of its place and dramatically growing its worth as soon as there isn’t any extra competitors and shoppers haven’t any selection however to buy the now high-priced drug. In these instances, it ought to be handled as the abuse that it’s and prosecuted the place acceptable.
Prosecuting such monopoly abuses might require new authority for the Federal Trade Commission (FTC). The FTC’s mission is “to prevent business practices that are anticompetitive or deceptive or unfair to consumers.” The FTC notes that it has restricted authority to take motion in opposition to an organization that has drastically raised the worth of a drug, relying on the motive for the improve.
Medicare Part D Reform
On a constructive notice, the BBBA takes a step in the proper path on drug coverage by realigning incentives in the Part D program—and Congress ought to work to enact reforms alongside these traces. While the construction of the BBBA’s Part D reform is essentially the identical as AAF’s authentic proposal—in addition to House Republican’s proposal in H.R. 19 and the proposal included in H.R. 3—there are some variations. AAF thought-about capping beneficiary OOP prices between $2,500 and $4,000 yearly. The BBBA caps OOP prices at $2,000, considerably under what beneficiaries are anticipated to pay earlier than transferring into the catastrophic part beneath present regulation. One potential enchancment, recognizing budgetary constraints and the must stability financial savings for beneficiaries with prices for taxpayers, could be a barely larger OOP. This could possibly be coupled with a discount in beneficiary coinsurance under the cap, which might profit extra enrollees—since most won’t ever attain the OOP cap—whereas nonetheless offering substantial financial savings for taxpayers and enrollees who do attain the cap.
Conclusion
The BBBA’s drug pricing provisions have been bemoaned by progressives as far quick of what is important, and touted by reasonable Democrats as extra affordable and fewer draconian than previous efforts comparable to H.R. 3. Both these views are essentially flawed. The BBBA would make large-scale modifications to drug coverage at the federal degree and attain deep into non-public insurance coverage and contracts. These insurance policies would have widespread, unfavourable impacts on the growth of future therapies, new indications for present therapies, and the financial advantages the United States derives from a vibrant biopharmaceutical sector. If the BBBA’s insurance policies are enacted in totality, American sufferers will undergo, American management in medical analysis will probably be diminished, and a vibrant engine of financial growth for American staff and buyers will probably be strangled.
Notes
[1] https://www.healthaffairs.org/doi/10.1377/hlthaff.2021.01763
[2] https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/NationalHealthExpendData/NationalHealthAccountsHistorical
[3] https://altarum.org/sites/default/files/uploaded-publication-files/Altarum%20Projections%20of%20the%20Non-Retail%20Dru.pdf
[4] https://www.americanactionforum.org/testimony/testimony-on-the-lower-drug-costs-now-act-h-r-3/
[5] https://www.americanactionforum.org/research/key-health-policy-provisions-of-the-build-back-better-act/
[6] https://go.bio.org/rs/490-EHZ-999/images/ClinicalDevelopmentSuccessRates2011_2020.pdf?_ga=2.112327436.987275036.1641911607-1139759599.1641911607
[7] https://cpb-us-w2.wpmucdn.com/voices.uchicago.edu/dist/d/3128/files/2021/08/Issue-Brief-Drug-Pricing-in-HR-5376-11.30.pdf
[8] https://www.americanactionforum.org/comments-for-record/comments-to-cms-on-proposed-international-pricing-index-for-medicare-part-b-drugs/
[9] https://www.fda.gov/drugs/new-drugs-fda-cders-new-molecular-entities-and-new-therapeutic-biological-products/novel-drug-approvals-2021
[10] https://www.americanactionforum.org/research/competition-and-the-medicare-part-d-program/
[11] https://www.kff.org/medicare/issue-brief/medicare-part-d-a-first-look-at-medicare-prescription-drug-plans-in-2022/
[12] https://www.cbo.gov/sites/default/files/108th-congress-2003-2004/reports/fristletter.pdf
[13] https://www.nber.org/papers/w12676
[14] https://www.abpi.org.uk/facts-and-figures/science-and-innovation/worldwide-pharmaceutical-company-rd-expenditure-by-country/
[15] https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/Information-on-Prescription-Drugs/index.html
[16] https://www.fda.gov/NewsEvents/Newsroom/FDAInBrief/ucm625627.htm